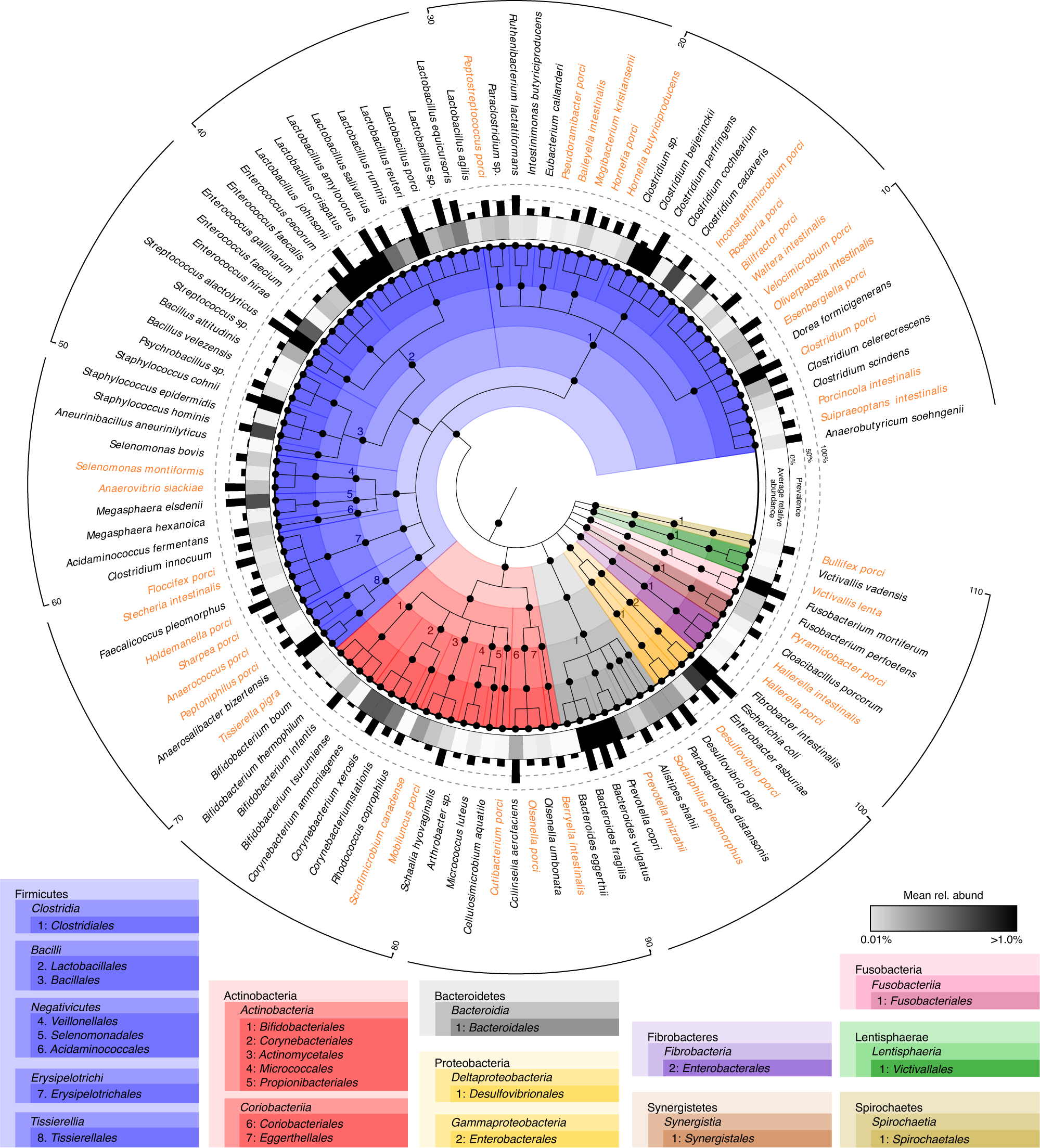
A collection of bacterial isolates from the pig intestine reveals functional and taxonomic diversity | Nature Communications

Omni's Dallas designer Waldrop + Nichols Studio opens up about its Louisville-centric approach - Louisville Business First

Environment-Driven Coherent Population Transfer Governs the Ultrafast Photophysics of Tryptophan | Journal of the American Chemical Society
![Discovery of 1-(4-Methoxyphenyl)-7-oxo-6-(4-(2-oxopiperidin-1-yl)phenyl)-4,5,6,7-tetrahydro- 1H-pyrazolo[3,4-c]pyridine-3-carboxamide (Apixaban, BMS-562247), a Highly Potent, Selective, Efficacious, and Orally Bioavailable Inhibitor of Blood ... Discovery of 1-(4-Methoxyphenyl)-7-oxo-6-(4-(2-oxopiperidin-1-yl)phenyl)-4,5,6,7-tetrahydro- 1H-pyrazolo[3,4-c]pyridine-3-carboxamide (Apixaban, BMS-562247), a Highly Potent, Selective, Efficacious, and Orally Bioavailable Inhibitor of Blood ...](https://pubs.acs.org/cms/10.1021/jm070245n/asset/images/large/jm070245nn00001.jpeg)
Discovery of 1-(4-Methoxyphenyl)-7-oxo-6-(4-(2-oxopiperidin-1-yl)phenyl)-4,5,6,7-tetrahydro- 1H-pyrazolo[3,4-c]pyridine-3-carboxamide (Apixaban, BMS-562247), a Highly Potent, Selective, Efficacious, and Orally Bioavailable Inhibitor of Blood ...
![Inclusion of Pentamidine in Carboxylated Pillar[5]arene: Late Sequential Crystallization and Diversity of Host–Guest Interactions | Crystal Growth & Design Inclusion of Pentamidine in Carboxylated Pillar[5]arene: Late Sequential Crystallization and Diversity of Host–Guest Interactions | Crystal Growth & Design](https://pubs.acs.org/cms/10.1021/acs.cgd.1c01201/asset/images/acs.cgd.1c01201.social.jpeg_v03)
Inclusion of Pentamidine in Carboxylated Pillar[5]arene: Late Sequential Crystallization and Diversity of Host–Guest Interactions | Crystal Growth & Design



















